MECHANISM · STUDIES · CITATIONS
The Science of AllaQuix®
How chitosan works independently of the clotting cascade.
A technical overview of AllaQuix® High-Performance Stop Bleeding Gauze™: the chitosan mechanism of action, peer-reviewed studies, safety profile, and clinical applications. For clinicians, wound-care specialists, and patients who want the science behind the product.
In this overview: Mechanism · Advantages · Safety · References

ABOUT ALLAQUIX®
Professional-grade hemostatic dressing
AllaQuix® High-Performance Stop Bleeding Gauze™ is a sterile, topical, active hemostatic wound dressing. FDA reviewed and cleared. Designed for management of minor cuts, abrasions, and lacerations.
- Sterile, drug-free, hypoallergenic, plant-based
- Works independently of the clotting cascade
- Made with chitosan, the same technology used by the U.S. Military
- FDA reviewed and cleared (510(k) medical device)
- Available over the counter, without a prescription
CHITOSAN
A nearly five-decade history in medicine
Chitosan has a long history as a hemostat. Early work on using chitosan in medical applications, specifically for hemostasis, dates back nearly five decades. It is a natural polysaccharide derived from the partial deacetylation of chitin, the structural component of crustacean shells, insect exoskeletons, and fungal cell walls.
~50 yrs of medical and surgical research literature · 20+ yrs in field use by the U.S. Military · Natural chitin-derived polysaccharide
MECHANISM OF ACTION
How chitosan stops bleeding
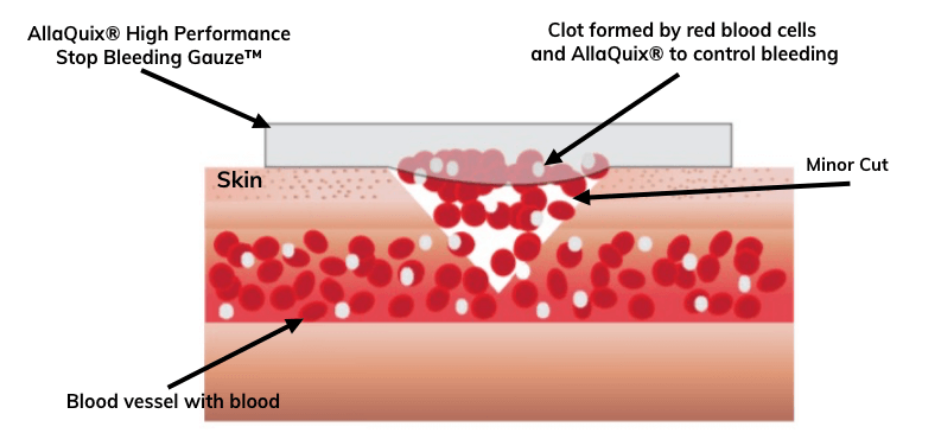
Hemostasis induced by chitosan is attributed to the physical interaction between chitosan and the cell membranes of red blood cells. The positively-charged chitosan molecule and the negatively-charged cell membrane form a stable coagulum independently of the normal coagulation pathway.
Charge interaction: Chitosan (+) attracts red blood cells (−), forming a mechanical seal independent of the clotting cascade.

Micrograph
Heparinized blood forms a bond with chitosan fibers to form a stable coagulum. Results replicable across studies.
How the seal forms
Chitosan's positive charge attracts negatively charged red blood cells into a mechanical seal at the wound surface, independent of the clotting cascade, which is why it can work for people who normally have trouble stopping bleeding.
Advantages of AllaQuix® High-Performance
Safety of AllaQuix®
-
What AllaQuix® is
- Sterilized through gamma irradiation
- FDA reviewed and cleared (510(k) device)
- No contraindications
- No age restrictions
- Sterile, drug-free, hypoallergenic, plant-based
-
What AllaQuix® is not for
- Medical emergencies
- Serious or arterial wounds
- Persistent uncontrolled bleeding
- Replacement for clinical advice or stitches when stitches are needed
- Internal use
Note on shellfish allergies
AllaQuix® is made from chitosan, which is derived from shellfish. Individuals with known shellfish allergies should exercise caution. A study published in Military Medicine evaluating "Safety of Chitosan Bandages in Shellfish Allergic Patients" is summarized in the references below.
CITATIONS
Scientific references
- Acheson EM, Kheirabadi BS, Deguzman R, Dick EJ Jr, Holcomb JB. Comparison of hemorrhagic control agents applied to lethal extremity arterial hemorrhages in swine. J Trauma. 2005.
- Brown MA, Daya MR, Worley JA. Experience with chitosan dressings in a civilian EMS system. J Emerg Med. 2009.
- Kheirabadi BS, et al. Safety evaluation of new hemostatic agents, smectite granules, and kaolin-coated gauze in a vascular injury wound model in swine. J Trauma. 2010.
- Wedmore I, McManus JG, Pusateri AE, Holcomb JB. A special report on the chitosan-based hemostatic dressing: experience in current combat operations. J Trauma. 2006.
- Pusateri AE, McCarthy SJ, Gregory KW, et al. Effect of a chitosan-based hemostatic dressing on blood loss and survival in a model of severe venous hemorrhage and hepatic injury in swine. J Trauma. 2003.
- Waibel KH, Haney B, Moore M, Whisman B, Gomez R. Safety of chitosan bandages in shellfish allergic patients. Mil Med. 2011.
TRY ALLAQUIX®
Try the science for yourself
Not for emergency, arterial, or severe bleeding.
